|
(iv) Determination of charge only due to uniform magnetic field. Substituting the values on RHS, the value of specific charge is calculated as Where C is proportionality constant which depends on the geometry of the discharge tube and substituting y. Therefore, the deflection y on the screen is of cathode rays is (note: u = 0 and a e = e m E) Let l be the length of one of the plates, then the time taken is Let t be the time taken by the cathode rays to travel in electric field. Let the initial upward velocity on the screen.Let the initial upward velocity of cathode ray be u = 0 before entering the parallel electric plates. Let y be the deviation produced from original position on the screen. Substituting equation (4) in equation (3), 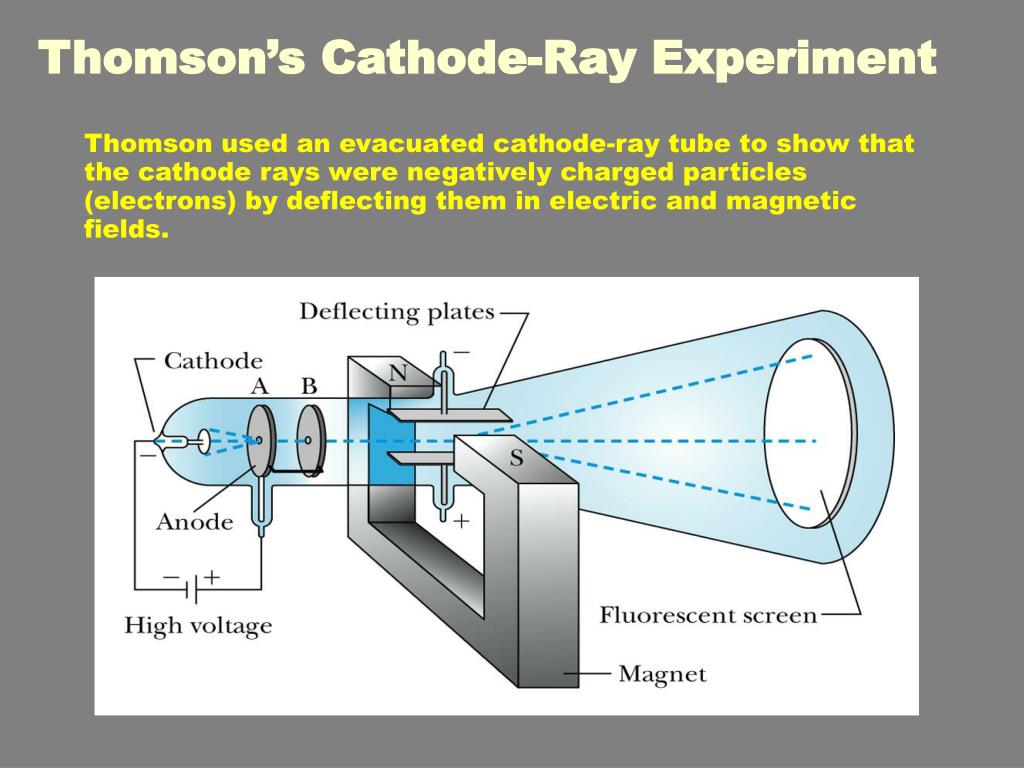
Let m be the mass of the electron and by applying Newton.s second law of motion,acceleration of the electron is The deflection in vertical direction is due to the electric force. When the magnetic field is turned off, the deflection is only due to electric field. (iii)Determination of charge only due to uniform electric field Substituting the values of E,B and V, the specific charge can be determined as Substituting the value of velocity from equation (1), we get Let V be the potential difference between anode and cathode, then the potential energy is eV. Since the cathode rays (electron beam) are accelerated from cathode to anode, the potential energy of the electron beam at the cathode is converted into kinetic energy of the electron beam at the anode. Lete be the charge of the cathode rays, then (i) Determination of velocity of cathode raysįor a fixed electric field between the plates, the magnetic field is adjusted such that the cathode rays(electron beam) strike at the original position O.This means that the magnitude of electric force is balanced by the magnitude of force due to magnetic field. This is achieved by coating the screen with zinc sulphide. These cathode rays are now allowed to pass through the parallel metal plates, maintained at high voltage.Further this gas discharge tube is kept in between pole pieces of magnet such that both electric and magnetic fields are perpendicular to each other.When the cathode rays strike the screen, they produce scintillation and hence bright spot is observed. Anode disc is made with pin hole in order to allow only a narrow beam of cathode rays. By the variation of electric and magnetic fields, mass normalized charge or the specific charge(charge per unit mass) of the cathode rays is measured.Ī highly evacuated discharge tube is used and cathode rays (electron beam) produced at cathode are attracted towards anode disc A. In the presence of electric and magnetic fields, the cathode rays are deflected. Everett who helped to greatly increase Thomson's experimental range.In 1887, J.J.Thomson made remarkable improvement in the scope of study of gases in discharge tubes. About 1894 he acquired an excellent glassblower named E. He was very fumble fingered and had a tendancy to break things. Incidently, Thomson was a very unhandy person. The amount the cathode ray bent from the straight line using either the electric field or the magnetic field allowed Thomson to calculate the e/m ratio. This allowed him to use either electrical or magnetic or a combination of both to cause the cathode ray to bend.  
Thomson also could use magnets, which were placed on either side of the straight portion of the tube just to the right of the electrical plates. The two plates about midway in the CRT were connected to a powerful electric battery thereby creating a strong electrical field through which the cathode rays passed. The long glass finger (in the photo) projecting downward from the right-hand globe is where the entire tube was evacuated down to as good as a vacuum as could be produced, then sealed. Thomson in 1897 announcing the discovery of the electron. Th diagram below appeared in an article by J.J. It is about one meter in length and was made entirely by hand. The image below of a CRT used by Thomson in his experiments. Only the end of the CRT can be seen to the right-hand side of the picture. Thomson and a cathode ray tube from around 1897, the year he announced the discovery of the electron. Thomson used results from cathode ray tube (commonly abbreviated CRT) experiments to discover the electron.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
